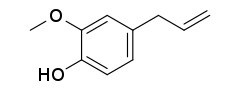
Eugenol
| |

| |
| Names | |
|---|---|
| Preferred IUPAC name
2-Methoxy-4-(prop-2-en-1-yl)phenol | |
Other names
| |
| Identifiers | |
3D model (JSmol)
|
|
| 1366759 | |
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.002.355 |
| EC Number |
|
| KEGG | |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C10H12O2 | |
| Molar mass | 164.204 g·mol−1 |
| Density | 1.06 g/cm3 |
| Melting point | −7.5 °C (18.5 °F; 265.6 K) |
| Boiling point | 254 °C (489 °F; 527 K) |
| Acidity (pKa) | 10.19 at 25 °C |
| −1.021×10−4 cm3/mol | |
| Viscosity |
|
| Hazards | |
| NFPA 704 (fire diamond) | |
| Flash point | 104 °C (219 °F; 377 K) |
| Related compounds | |
Related compounds
|
2-Phenethyl propionate |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Eugenol /ˈjuːdʒɪnɒl/ is an allyl chain-substituted guaiacol, a member of the allylbenzene class of chemical compounds.[2] It is a colorless to pale yellow, aromatic oily liquid extracted from certain essential oils especially from clove, nutmeg, cinnamon, basil and bay leaf.[3][4][5][6] It is present in concentrations of 80–90% in clove bud oil and at 82–88% in clove leaf oil.[7] Eugenol has a pleasant, spicy, clove-like scent.[8] The name is derived from Eugenia caryophyllata, the former Linnean nomenclature term for cloves. The currently accepted name is Syzygium aromaticum.[9]
Biosynthesis
[edit]The biosynthesis of eugenol begins with the amino acid tyrosine. L-tyrosine is converted to p-coumaric acid by the enzyme tyrosine ammonia lyase (TAL).[10] From here, p-coumaric acid is converted to caffeic acid by p-coumarate 3-hydroxylase using oxygen and NADPH. S-Adenosyl methionine (SAM) is then used to methylate caffeic acid, forming ferulic acid, which is in turn converted to feruloyl-CoA by the enzyme 4-hydroxycinnamoyl-CoA ligase (4CL).[11] Next, feruloyl-CoA is reduced to coniferaldehyde by cinnamoyl-CoA reductase (CCR). Coniferaldeyhyde is then further reduced to coniferyl alcohol by cinnamyl-alcohol dehydrogenase (CAD) or sinapyl-alcohol dehydrogenase (SAD). Coniferyl alcohol is then converted to an ester in the presence of the substrate CH3COSCoA, forming coniferyl acetate. Finally, coniferyl acetate is converted to eugenol via the enzyme eugenol synthase 1 and the use of NADPH.[citation needed]
Eugenol is a metabolite of Caleicine, the active compound found in Calea Ternifolia, and is thought to cause the sedative and hallucinogenic state Calea Ternifolia can induce.

Pharmacology
[edit]Eugenol and thymol possess general anesthetic properties. Like many other anesthetic agents, these 2-alkyl(oxy)phenols act as positive allosteric modulators of the GABAA receptor. Although eugenol and thymol are too toxic and not potent enough to be used clinically, these findings led to the development of 2-substituted phenol anesthetic drugs, including propanidid (later withdrawn) and the widely used propofol.[12] Eugenol and the structurally similar myristicin, have the common property of inhibiting MAO-A and MAO-B in vitro.[13][14]
Eugenol acts on the NDMA receptors as an antagonist, as well as the histamine receptors as an antagonist, but the exact specifics of this are unknown. [15]
In humans, complete excretion occurs within 24 hour and metabolites are mostly conjugates of eugenol.[16]
Uses
[edit]Humans
[edit]Eugenol is used as a flavor or aroma ingredient in teas, meats, cakes, perfumes, cosmetics, flavorings, and essential oils.[2][17][18] It is also used as a local antiseptic and anaesthetic.[19][20] Eugenol can be combined with zinc oxide to form zinc oxide eugenol which has restorative and prosthodontic applications in dentistry. For persons with a dry socket as a complication of tooth extraction, packing the dry socket with a eugenol-zinc oxide paste on iodoform gauze is effective for reducing acute pain.[21] Eugenol-zinc oxide paste is also used for root canal sealing.[22]
Insects and fish
[edit]It is attractive to males of various species of orchid bees, which apparently gather the chemical to synthesize pheromones; it is commonly used as bait to attract and collect these bees for study.[23] It also attracts female cucumber beetles.[24]
Eugenol and isoeugenol, which both are floral volatile scent compounds, are catalyzed by a single type of enzyme in the genus Gymnadenia and the gene encoding for this enzyme is the first functionally characterized gene in these species.[25] Eugenol is an ingredient in some insecticides.[2]
Clove oil is common as an anesthetic for use on aquarium fish as well as on wild fish when sampled for research and management purposes.[26][27] Where readily available, it presents a humane method to euthanize sick and diseased fish either by direct overdose or to induce sleep before an overdose of eugenol.[28][29]
Other
[edit]Eugenol is an ingredient in some fungicides and weed control products used in agricultural practices in the European Union.[2] It is used in hundreds of household products, such as pesticides, pet care, laundry, cleaning, and paper or vehicle products.[2]
Toxicity
[edit]Lesser side effects of eugenol toxicity that may not be considered a full overdose: Sedation, dizziness, hallucinations, mild respiratory depression, nausea, and muscle spasms.
Taken orally in high doses for chronic periods, eugenol may cause liver toxicity.[17] An overdose is possible, causing a wide range of symptoms, such as hematuria (blood in urine), convulsions, diarrhea, delirium, unconsciousness, heavy respiratory depression, tachycardia (rapid heart rate), or acute kidney injury.[17][30][31] N-acetylcysteine may be used to treat people with eugenol or clove oil overdose.[32]
As an allergenic
[edit]Eugenol is subject to restrictions on its use in perfumery,[33] as some people may become sensitised to it, however, the degree to which eugenol can cause an allergic reaction in humans is disputed.[34]
Eugenol is a component of balsam of Peru, to which some people are allergic.[35][36] When eugenol is used in dental preparations such as surgical pastes, dental packing, and dental cement, it may cause contact stomatitis and allergic cheilitis.[35] The allergy can be discovered via a patch test.[35]
Natural occurrence
[edit]Eugenol naturally occurs in numerous plants, including the following:
- Cloves (Syzygium aromaticum)[37][38][39]
- Wormwood[citation needed]
- Cinnamon[38][40]
- Cinnamomum tamala[41]
- Nutmeg (Myristica fragrans)[42]
- Ocimum basilicum (sweet basil)[43]
- Ocimum gratissimum (African basil)[25][44]
- Ocimum tenuiflorum (syn. Ocimum sanctum, tulsi or holy basil)
- Japanese star anise[45]
- Lemon balm[46]
- Dill[citation needed]
- Pimenta dioica (Allspice) [citation needed]
- Vanilla[citation needed]
- Bay laurel[citation needed]
- Celery[citation needed]
- Ginger[citation needed]
See also
[edit]References
[edit]- ^ Bingham EC, Spooner LW (1932). "The Fluidity Method for the Determination of Association. I". Journal of Rheology. 3 (2): 221–244. Bibcode:1932JRheo...3..221B. doi:10.1122/1.2116455. ISSN 0097-0360.
- ^ a b c d e "Eugenol". PubChem, US National Library of Medicine. 16 October 2021. Retrieved 24 October 2021.
- ^ "Constituents of the essential oil from leaves and buds of clove (Syzigium caryophyllatum L.) Alston" (PDF). Bangladesh Council of Scientific and Industrial Research BCSIR Laboratories. 4: 451–454.
- ^ Mallavarapu GR, Ramesh S, Chandrasekhara RS, Rajeswara Rao BR, Kaul PN, Bhattacharya AK (1995). "Investigation of the essential oil of cinnamon leaf grown at Bangalore and Hyderabad". Flavour and Fragrance Journal. 10 (4): 239–242. doi:10.1002/ffj.2730100403.
- ^ Yield and Oil Composition of 38 Basil (Ocimum basilicum L.) Accessions Grown in Mississippi Archived 15 October 2010 at the Wayback Machine
- ^ "Typical G.C. for bay leaf oil". Thegoodscentscompany.com. Archived from the original on 17 March 2014. Retrieved 27 April 2014.
- ^ Barnes J, Anderson LA, Phillipson JS (2007) [1996]. Herbal Medicines (PDF) (3rd ed.). London: Pharmaceutical Press. ISBN 978-0-85369-623-0. Archived from the original (PDF) on 1 July 2018. Retrieved 27 April 2015.
- ^ Wishart, David S.; Guo, An Chi; Oler, Eponine; et al. "Showing metabocard for Eugenol (HMDB0005809)". Human Metabolome Database, HMDB. 5.0.
- ^ Cortés Rojas DF, de Souza CR, Oliveira WP (February 2014). "Clove (Syzygium aromaticum): a precious spice". Asian Pacific Journal of Tropical Biomedicine. 4 (2): 90–6. doi:10.1016/S2221-1691(14)60215-X. PMC 3819475. PMID 25182278.
- ^ Dewick, P. M. (2009). Medicinal Natural Products. John Wiley & Sons. doi:10.1002/9780470742761. ISBN 9780470742761.
- ^ Harakava, R. (2005). "Genes encoding enzymes of the lignin biosynthesis pathway in Eucalyptus". Genet. Mol. Biol. 28 (3 suppl): 601–607. doi:10.1590/S1415-47572005000400015.
- ^ Tsuchiya H (August 2017). "Anesthetic Agents of Plant Origin: A Review of Phytochemicals with Anesthetic Activity". Molecules. 22 (8): 1369. doi:10.3390/molecules22081369. PMC 6152143. PMID 28820497.
- ^ Tao G, Irie Y, Li DJ, Keung WM (August 2005). "Eugenol and its structural analogs inhibit monoamine oxidase A and exhibit antidepressant-like activity". Bioorganic & Medicinal Chemistry. 13 (15): 4777–88. doi:10.1016/j.bmc.2005.04.081. PMID 15936201.
- ^ Tisserand, Robert; Young, Rodney (2013). Essential oil safety : a guide for health care professionals (Second ed.). Edinburgh. p. 73. ISBN 978-0443062414. Archived from the original on 17 December 2020.
{{cite book}}: CS1 maint: location missing publisher (link) - ^ Nisar, Muhammad Farrukh; Khadim, Mahnoor; Rafiq, Muhammad; Chen, Jinyin; Yang, Yali; Wan, Chunpeng Craig (3 August 2021). "Pharmacological Properties and Health Benefits of Eugenol: A Comprehensive Review". Oxidative Medicine and Cellular Longevity. 2021: 2497354. doi:10.1155/2021/2497354. ISSN 1942-0900. PMC 8357497. PMID 34394824.
- ^ Fischer IU, von Unruh GE, Dengler HJ (February 1990). "The metabolism of eugenol in man". Xenobiotica; the Fate of Foreign Compounds in Biological Systems. 20 (2): 209–22. doi:10.3109/00498259009047156. PMID 2333717.
- ^ a b c "Eugenol (clove oil)". LiverTox, US National Institute of Diabetes and Digestive and Kidney Diseases. 28 October 2018. PMID 31869191. Retrieved 24 October 2021.
- ^ "Hair loss after Corona vaccination and infection: Chief doctor clarifies". www.colcorona.net. Retrieved 10 December 2022.
- ^ Sell AB, Carlini EA (1976). "Anesthetic action of methyleugenol and other eugenol derivatives". Pharmacology. 14 (4): 367–77. doi:10.1159/000136617. PMID 935250.
- ^ Jadhav BK, Khandelwal KR, Ketkar AR, Pisal SS (February 2004). "Formulation and evaluation of mucoadhesive tablets containing eugenol for the treatment of periodontal diseases". Drug Development and Industrial Pharmacy. 30 (2): 195–203. doi:10.1081/DDC-120028715. PMID 15089054. S2CID 19138725.
- ^ Tarakji B, Saleh LA, Umair A, Azzeghaiby SN, Hanouneh S (April 2015). "Systemic review of dry socket: aetiology, treatment, and prevention". Journal of Clinical and Diagnostic Research. 9 (4): ZE10-3. doi:10.7860/JCDR/2015/12422.5840. PMC 4437177. PMID 26023661.
- ^ Ferracane, Jack L. (2001). Materials in Dentistry: Principles and Applications (2nd ed.). Lippincott, Williams & Wilkins. ISBN 978-0-7817-2733-4.
- ^ Schiestl FP, Roubik DW (January 2003). "Odor compound detection in male euglossine bees". Journal of Chemical Ecology. 29 (1): 253–7. doi:10.1023/A:1021932131526. hdl:20.500.11850/57276. PMID 12647866. S2CID 2845587.
- ^ "Cucumber Beetles: Organic and Biorational Integrated Pest Management (Summary)". Attra.ncat.org. 5 August 2013. Archived from the original on 16 May 2010. Retrieved 27 April 2014.
- ^ a b Gupta AK, Schauvinhold I, Pichersky E, Schiestl FP (December 2014). "Eugenol synthase genes in floral scent variation in Gymnadenia species" (PDF). Functional & Integrative Genomics. 14 (4): 779–88. doi:10.1007/s10142-014-0397-9. hdl:20.500.11850/91540. PMID 25239559. S2CID 17207240.
- ^ Anesthesia, Analgesia, and Surgery in Pet Fish. Atlantic Coast Veterinary Conference. 2001. Archived from the original on 5 March 2009. Retrieved 17 March 2014.
- ^ Grush J, Noakes DL, Moccia RD (February 2004). "The efficacy of clove oil as an anesthetic for the zebrafish, Danio rerio (Hamilton)". Zebrafish. 1 (1): 46–53. doi:10.1089/154585404774101671. PMID 18248205.
- ^ Monks, Neale (2 April 2009). "Aquarium Fish Euthanasia" (PDF). Fish Channel. Archived from the original (PDF) on 26 July 2011. Retrieved 7 December 2010.
- ^ de Souza Valente, Cecília (June 2022). "Anaesthesia of decapod crustaceans". Veterinary and Animal Science. 16: 100252. doi:10.1016/j.vas.2022.100252. PMC 9127210. PMID 35620220.
- ^ "Eugenol Oil Overdose". The New York Times. Archived from the original on 25 July 2011.
- ^ "Eugenol (Clove Oil)", LiverTox: Clinical and Research Information on Drug-Induced Liver Injury, Bethesda (MD): National Institute of Diabetes and Digestive and Kidney Diseases, 2012, PMID 31869191, retrieved 20 May 2024
- ^ Janes, S. E.; Price, C. S.; Thomas, D. (2005). "Essential oil poisoning: N-acetylcysteine for eugenol-induced hepatic failure and analysis of a national database". European Journal of Pediatrics. 164 (8): 520–522. doi:10.1007/s00431-005-1692-1. PMID 15895251. S2CID 6452985.
- ^ "IFRA". www.ifraorg.org. Archived from the original on 30 December 2011.
- ^ "Cropwatch Claims Victory Regarding "26 Allergens" Legislation" (PDF). leffingwell.com. Archived from the original (PDF) on 10 February 2014. Retrieved 27 April 2014.
- ^ a b c Schmalz G, Bindslev DA (2008). Biocompatibility of Dental Materials. Springer. p. 352. ISBN 978-3-540-77782-3.
- ^ Bruynzeel, Derk P. (2014). "Balsam of Peru (Myroxylon pereirae)". Management of Positive Patch Test Reactions. Springer. pp. 53–55. doi:10.1007/978-3-642-55706-4_11. ISBN 978-3-540-44347-6.
- ^ Pathak SB, Niranjan K, Padh H, Rajani M (2004). "TLC Densitometric Method for the Quantification of Eugenol and Gallic Acid in Clove". Chromatographia. 60 (3–4): 241–244. doi:10.1365/s10337-004-0373-y. S2CID 95396304.
- ^ a b Bullerman LB, Lieu FY, Seier SA (July 1977). "Inhibition of growth and aflatoxin production by cinnamon and clove oils. Cinnamic aldehyde and eugenol". Journal of Food Science. 42 (4): 1107–1109. doi:10.1111/j.1365-2621.1977.tb12677.x.
- ^ Lee KG, Shibamoto T (2001). "Antioxidant property of aroma extract isolated from clove buds [Syzygium aromaticum (L.) Merr. et Perry]". Food Chemistry. 74 (4): 443–448. doi:10.1016/S0308-8146(01)00161-3.
- ^ Kreydiyyeh SI, Usta J, Copti R (September 2000). "Effect of cinnamon, clove and some of their constituents on the Na(+)-K(+)-ATPase activity and alanine absorption in the rat jejunum". Food and Chemical Toxicology. 38 (9): 755–62. doi:10.1016/S0278-6915(00)00073-9. PMID 10930696.
- ^ Dighe VV, Gursale AA, Sane RT, Menon S, Patel PH (2005). "Quantitative Determination of Eugenol from Cinnamomum tamala Nees and Eberm. Leaf Powder and Polyherbal Formulation Using Reverse Phase Liquid Chromatography". Chromatographia. 61 (9–10): 443–446. doi:10.1365/s10337-005-0527-6. S2CID 97399632.
- ^ Bennett A, Stamford IF, Tavares IA, Jacobs S, Capasso F, Mascolo N, et al. (1988). "The biological activity of eugenol, a major constituent of nutmeg (..Myristica fragrans..): Studies on prostaglandins, the intestine and other tissues". Phytotherapy Research. 2 (3): 124–130. doi:10.1002/ptr.2650020305. S2CID 85114864.
- ^ Johnson CB, Kirby J, Naxakis G, Pearson S (1999). "Substantial UV-B-mediated induction of essential oils in sweet basil (Ocimum basilicum L.)". Phytochemistry. 51 (4): 507–510. Bibcode:1999PChem..51..507J. doi:10.1016/S0031-9422(98)00767-5.
- ^ Nakamura CV, Ueda-Nakamura T, Bando E, Melo AF, Cortez DA, Dias Filho BP (September 1999). "Antibacterial activity of Ocimum gratissimum L. essential oil". Memórias do Instituto Oswaldo Cruz. 94 (5): 675–8. doi:10.1590/S0074-02761999000500022. PMID 10464416.
- ^ Ize-Ludlow D, Ragone S, Bruck IS, Bernstein JN, Duchowny M, Peña BM (November 2004). "Neurotoxicities in infants seen with the consumption of star anise tea". Pediatrics. 114 (5): e653-6. doi:10.1542/peds.2004-0058. PMID 15492355.
- ^ "Lemon balm". University of Maryland Medical Center. Archived from the original on 1 August 2013. Retrieved 7 December 2020.

