Gene expression
| Part of a series on |
| Biochemistry |
|---|
 |
Gene expression is the process by which information from a gene is used in the synthesis of a functional gene product that enables it to produce end products, proteins or non-coding RNA, and ultimately affect a phenotype. These products are often proteins, but in non-protein-coding genes such as transfer RNA (tRNA) and small nuclear RNA (snRNA), the product is a functional non-coding RNA. The process of gene expression is used by all known life—eukaryotes (including multicellular organisms), prokaryotes (bacteria and archaea), and utilized by viruses—to generate the macromolecular machinery for life.
In genetics, gene expression is the most fundamental level at which the genotype gives rise to the phenotype, i.e. observable trait. The genetic information stored in DNA represents the genotype, whereas the phenotype results from the "interpretation" of that information. Such phenotypes are often displayed by the synthesis of proteins that control the organism's structure and development, or that act as enzymes catalyzing specific metabolic pathways.
All steps in the gene expression process may be modulated (regulated), including the transcription, RNA splicing, translation, and post-translational modification of a protein. Regulation of gene expression gives control over the timing, location, and amount of a given gene product (protein or ncRNA) present in a cell and can have a profound effect on the cellular structure and function. Regulation of gene expression is the basis for cellular differentiation, development, morphogenesis and the versatility and adaptability of any organism. Gene regulation may therefore serve as a substrate for evolutionary change.
Mechanism
[edit]Transcription
[edit]
The production of a RNA copy from a DNA strand is called transcription, and is performed by RNA polymerases, which add one ribonucleotide at a time to a growing RNA strand as per the complementarity law of the nucleotide bases. This RNA is complementary to the template 3′ → 5′ DNA strand,[1] with the exception that thymines (T) are replaced with uracils (U) in the RNA and possible errors.
In bacteria, transcription is carried out by a single type of RNA polymerase, which needs to bind a DNA sequence called a Pribnow box with the help of the sigma factor protein (σ factor) to start transcription. In eukaryotes, transcription is performed in the nucleus by three types of RNA polymerases, each of which needs a special DNA sequence called the promoter and a set of DNA-binding proteins—transcription factors—to initiate the process (see regulation of transcription below). RNA polymerase I is responsible for transcription of ribosomal RNA (rRNA) genes. RNA polymerase II (Pol II) transcribes all protein-coding genes but also some non-coding RNAs (e.g., snRNAs, snoRNAs or long non-coding RNAs). RNA polymerase III transcribes 5S rRNA, transfer RNA (tRNA) genes, and some small non-coding RNAs (e.g., 7SK). Transcription ends when the polymerase encounters a sequence called the terminator.
mRNA processing
[edit]While transcription of prokaryotic protein-coding genes creates messenger RNA (mRNA) that is ready for translation into protein, transcription of eukaryotic genes leaves a primary transcript of RNA (pre-RNA), which first has to undergo a series of modifications to become a mature RNA. Types and steps involved in the maturation processes vary between coding and non-coding preRNAs; i.e. even though preRNA molecules for both mRNA and tRNA undergo splicing, the steps and machinery involved are different.[2] The processing of non-coding RNA is described below (non-coding RNA maturation).
The processing of pre-mRNA include 5′ capping, which is set of enzymatic reactions that add 7-methylguanosine (m7G) to the 5′ end of pre-mRNA and thus protect the RNA from degradation by exonucleases.[3] The m7G cap is then bound by cap binding complex heterodimer (CBC20/CBC80), which aids in mRNA export to cytoplasm and also protect the RNA from decapping.[4]
Another modification is 3′ cleavage and polyadenylation.[5] They occur if polyadenylation signal sequence (5′- AAUAAA-3′) is present in pre-mRNA, which is usually between protein-coding sequence and terminator.[6] The pre-mRNA is first cleaved and then a series of ~200 adenines (A) are added to form poly(A) tail, which protects the RNA from degradation.[7] The poly(A) tail is bound by multiple poly(A)-binding proteins (PABPs) necessary for mRNA export and translation re-initiation.[8] In the inverse process of deadenylation, poly(A) tails are shortened by the CCR4-Not 3′-5′ exonuclease, which often leads to full transcript decay.[9]
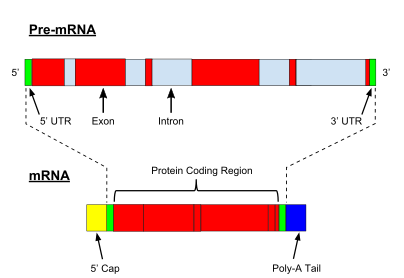
A very important modification of eukaryotic pre-mRNA is RNA splicing. The majority of eukaryotic pre-mRNAs consist of alternating segments called exons and introns.[10] During the process of splicing, an RNA-protein catalytical complex known as spliceosome catalyzes two transesterification reactions, which remove an intron and release it in form of lariat structure, and then splice neighbouring exons together.[11] In certain cases, some introns or exons can be either removed or retained in mature mRNA.[12] This so-called alternative splicing creates series of different transcripts originating from a single gene. Because these transcripts can be potentially translated into different proteins, splicing extends the complexity of eukaryotic gene expression and the size of a species proteome.[13]
Extensive RNA processing may be an evolutionary advantage made possible by the nucleus of eukaryotes. In prokaryotes, transcription and translation happen together, whilst in eukaryotes, the nuclear membrane separates the two processes, giving time for RNA processing to occur.[14]
Non-coding RNA maturation
[edit]In most organisms non-coding genes (ncRNA) are transcribed as precursors that undergo further processing. In the case of ribosomal RNAs (rRNA), they are often transcribed as a pre-rRNA that contains one or more rRNAs. The pre-rRNA is cleaved and modified (2′-O-methylation and pseudouridine formation) at specific sites by approximately 150 different small nucleolus-restricted RNA species, called snoRNAs. SnoRNAs associate with proteins, forming snoRNPs. While snoRNA part basepair with the target RNA and thus position the modification at a precise site, the protein part performs the catalytical reaction. In eukaryotes, in particular a snoRNP called RNase, MRP cleaves the 45S pre-rRNA into the 28S, 5.8S, and 18S rRNAs. The rRNA and RNA processing factors form large aggregates called the nucleolus.[15]
In the case of transfer RNA (tRNA), for example, the 5′ sequence is removed by RNase P,[16] whereas the 3′ end is removed by the tRNase Z enzyme[17] and the non-templated 3′ CCA tail is added by a nucleotidyl transferase.[18] In the case of micro RNA (miRNA), miRNAs are first transcribed as primary transcripts or pri-miRNA with a cap and poly-A tail and processed to short, 70-nucleotide stem-loop structures known as pre-miRNA in the cell nucleus by the enzymes Drosha and Pasha. After being exported, it is then processed to mature miRNAs in the cytoplasm by interaction with the endonuclease Dicer, which also initiates the formation of the RNA-induced silencing complex (RISC), composed of the Argonaute protein.
Even snRNAs and snoRNAs themselves undergo series of modification before they become part of functional RNP complex.[19] This is done either in the nucleoplasm or in the specialized compartments called Cajal bodies.[20] Their bases are methylated or pseudouridinilated by a group of small Cajal body-specific RNAs (scaRNAs), which are structurally similar to snoRNAs.[21]
RNA export
[edit]In eukaryotes most mature RNA must be exported to the cytoplasm from the nucleus. While some RNAs function in the nucleus, many RNAs are transported through the nuclear pores and into the cytosol.[22] Export of RNAs requires association with specific proteins known as exportins. Specific exportin molecules are responsible for the export of a given RNA type. mRNA transport also requires the correct association with Exon Junction Complex (EJC), which ensures that correct processing of the mRNA is completed before export. In some cases RNAs are additionally transported to a specific part of the cytoplasm, such as a synapse; they are then towed by motor proteins that bind through linker proteins to specific sequences (called "zipcodes") on the RNA.[23]
Translation
[edit]For some non-coding RNA, the mature RNA is the final gene product.[24] In the case of messenger RNA (mRNA) the RNA is an information carrier coding for the synthesis of one or more proteins. mRNA carrying a single protein sequence (common in eukaryotes) is monocistronic whilst mRNA carrying multiple protein sequences (common in prokaryotes) is known as polycistronic.
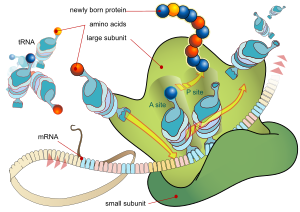
Every mRNA consists of three parts: a 5′ untranslated region (5′UTR), a protein-coding region or open reading frame (ORF), and a 3′ untranslated region (3′UTR). The coding region carries information for protein synthesis encoded by the genetic code to form triplets. Each triplet of nucleotides of the coding region is called a codon and corresponds to a binding site complementary to an anticodon triplet in transfer RNA. Transfer RNAs with the same anticodon sequence always carry an identical type of amino acid. Amino acids are then chained together by the ribosome according to the order of triplets in the coding region. The ribosome helps transfer RNA to bind to messenger RNA and takes the amino acid from each transfer RNA and makes a structure-less protein out of it.[25][26] Each mRNA molecule is translated into many protein molecules, on average ~2800 in mammals.[27][28]
In prokaryotes translation generally occurs at the point of transcription (co-transcriptionally), often using a messenger RNA that is still in the process of being created. In eukaryotes translation can occur in a variety of regions of the cell depending on where the protein being written is supposed to be. Major locations are the cytoplasm for soluble cytoplasmic proteins and the membrane of the endoplasmic reticulum for proteins that are for export from the cell or insertion into a cell membrane. Proteins that are supposed to be produced at the endoplasmic reticulum are recognised part-way through the translation process. This is governed by the signal recognition particle—a protein that binds to the ribosome and directs it to the endoplasmic reticulum when it finds a signal peptide on the growing (nascent) amino acid chain.[29]
Folding
[edit]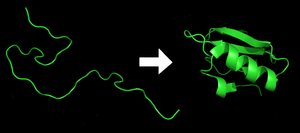
Each protein exists as an unfolded polypeptide or random coil when translated from a sequence of mRNA into a linear chain of amino acids. This polypeptide lacks any developed three-dimensional structure (the left hand side of the neighboring figure). The polypeptide then folds into its characteristic and functional three-dimensional structure from a random coil.[30] Amino acids interact with each other to produce a well-defined three-dimensional structure, the folded protein (the right hand side of the figure) known as the native state. The resulting three-dimensional structure is determined by the amino acid sequence (Anfinsen's dogma).[31]
The correct three-dimensional structure is essential to function, although some parts of functional proteins may remain unfolded.[32] Failure to fold into the intended shape usually produces inactive proteins with different properties including toxic prions. Several neurodegenerative and other diseases are believed to result from the accumulation of misfolded proteins.[33] Many allergies are caused by the folding of the proteins, for the immune system does not produce antibodies for certain protein structures.[34]
Enzymes called chaperones assist the newly formed protein to attain (fold into) the 3-dimensional structure it needs to function.[35] Similarly, RNA chaperones help RNAs attain their functional shapes.[36] Assisting protein folding is one of the main roles of the endoplasmic reticulum in eukaryotes.
Translocation
[edit]Secretory proteins of eukaryotes or prokaryotes must be translocated to enter the secretory pathway. Newly synthesized proteins are directed to the eukaryotic Sec61 or prokaryotic SecYEG translocation channel by signal peptides. The efficiency of protein secretion in eukaryotes is very dependent on the signal peptide which has been used.[37]
Protein transport
[edit]Many proteins are destined for other parts of the cell than the cytosol and a wide range of signalling sequences or (signal peptides) are used to direct proteins to where they are supposed to be.[38][39] In prokaryotes this is normally a simple process due to limited compartmentalisation of the cell.[40] However, in eukaryotes there is a great variety of different targeting processes to ensure the protein arrives at the correct organelle.[41]
Not all proteins remain within the cell and many are exported, for example, digestive enzymes, hormones and extracellular matrix proteins. In eukaryotes the export pathway is well developed and the main mechanism for the export of these proteins is translocation to the endoplasmic reticulum, followed by transport via the Golgi apparatus.[42][43]
Regulation of gene expression
[edit]
Regulation of gene expression is the control of the amount and timing of appearance of the functional product of a gene. Control of expression is vital to allow a cell to produce the gene products it needs when it needs them; in turn, this gives cells the flexibility to adapt to a variable environment, external signals, damage to the cell, and other stimuli. More generally, gene regulation gives the cell control over all structure and function, and is the basis for cellular differentiation, morphogenesis and the versatility and adaptability of any organism.
Numerous terms are used to describe types of genes depending on how they are regulated; these include:
- A constitutive gene is a gene that is transcribed continually as opposed to a facultative gene, which is only transcribed when needed.
- A housekeeping gene is a gene that is required to maintain basic cellular function and so is typically expressed in all cell types of an organism. Examples include actin, GAPDH and ubiquitin. Some housekeeping genes are transcribed at a relatively constant rate and these genes can be used as a reference point in experiments to measure the expression rates of other genes.
- A facultative gene is a gene only transcribed when needed as opposed to a constitutive gene.
- An inducible gene is a gene whose expression is either responsive to environmental change or dependent on the position in the cell cycle.
Any step of gene expression may be modulated, from the DNA-RNA transcription step to post-translational modification of a protein. The stability of the final gene product, whether it is RNA or protein, also contributes to the expression level of the gene—an unstable product results in a low expression level. In general gene expression is regulated through changes[44] in the number and type of interactions between molecules[45] that collectively influence transcription of DNA[46] and translation of RNA.[47]
Some simple examples of where gene expression is important are:
- Control of insulin expression so it gives a signal for blood glucose regulation.
- X chromosome inactivation in female mammals to prevent an "overdose" of the genes it contains.
- Cyclin expression levels control progression through the eukaryotic cell cycle.
Transcriptional regulation
[edit]
Regulation of transcription can be broken down into three main routes of influence; genetic (direct interaction of a control factor with the gene), modulation interaction of a control factor with the transcription machinery and epigenetic (non-sequence changes in DNA structure that influence transcription).[48][49]

Direct interaction with DNA is the simplest and the most direct method by which a protein changes transcription levels.[50] Genes often have several protein binding sites around the coding region with the specific function of regulating transcription.[51] There are many classes of regulatory DNA binding sites known as enhancers, insulators and silencers.[52] The mechanisms for regulating transcription are varied, from blocking key binding sites on the DNA for RNA polymerase to acting as an activator and promoting transcription by assisting RNA polymerase binding.[53]
The activity of transcription factors is further modulated by intracellular signals causing protein post-translational modification including phosphorylation, acetylation, or glycosylation.[54] These changes influence a transcription factor's ability to bind, directly or indirectly, to promoter DNA, to recruit RNA polymerase, or to favor elongation of a newly synthesized RNA molecule.[55]
The nuclear membrane in eukaryotes allows further regulation of transcription factors by the duration of their presence in the nucleus, which is regulated by reversible changes in their structure and by binding of other proteins.[56] Environmental stimuli or endocrine signals[57] may cause modification of regulatory proteins[58] eliciting cascades of intracellular signals,[59] which result in regulation of gene expression.
It has become apparent that there is a significant influence of non-DNA-sequence specific effects on transcription.[60] These effects are referred to as epigenetic and involve the higher order structure of DNA, non-sequence specific DNA binding proteins and chemical modification of DNA.[61] In general epigenetic effects alter the accessibility of DNA to proteins and so modulate transcription.[62]

In eukaryotes the structure of chromatin, controlled by the histone code, regulates access to DNA with significant impacts on the expression of genes in euchromatin and heterochromatin areas.[63]
Enhancers, transcription factors, mediator complex and DNA loops in mammalian transcription
[edit]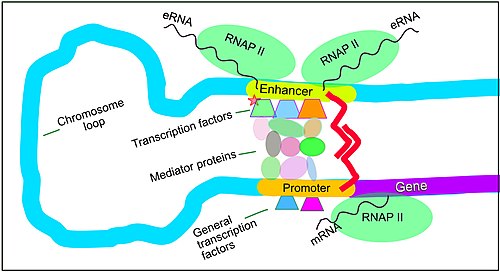
Gene expression in mammals is regulated by many cis-regulatory elements, including core promoters and promoter-proximal elements that are located near the transcription start sites of genes, upstream on the DNA (towards the 5' region of the sense strand). Other important cis-regulatory modules are localized in DNA regions that are distant from the transcription start sites. These include enhancers, silencers, insulators and tethering elements.[64] Enhancers and their associated transcription factors have a leading role in the regulation of gene expression.[65]
Enhancers are genome regions that regulate genes. Enhancers control cell-type-specific gene expression programs, most often by looping through long distances to come in physical proximity with the promoters of their target genes.[66] Multiple enhancers, each often tens or hundred of thousands of nucleotides distant from their target genes, loop to their target gene promoters and coordinate with each other to control gene expression.[66]
The illustration shows an enhancer looping around to come into proximity with the promoter of a target gene. The loop is stabilized by a dimer of a connector protein (e.g. dimer of CTCF or YY1). One member of the dimer is anchored to its binding motif on the enhancer and the other member is anchored to its binding motif on the promoter (represented by the red zigzags in the illustration).[67] Several cell function-specific transcription factors (among the about 1,600 transcription factors in a human cell)[68] generally bind to specific motifs on an enhancer.[69] A small combination of these enhancer-bound transcription factors, when brought close to a promoter by a DNA loop, govern transcription level of the target gene. Mediator (a complex usually consisting of about 26 proteins in an interacting structure) communicates regulatory signals from enhancer DNA-bound transcription factors directly to the RNA polymerase II (pol II) enzyme bound to the promoter.[70]
Enhancers, when active, are generally transcribed from both strands of DNA with RNA polymerases acting in two different directions, producing two eRNAs as illustrated in the figure.[71] An inactive enhancer may be bound by an inactive transcription factor. Phosphorylation of the transcription factor may activate it and that activated transcription factor may then activate the enhancer to which it is bound (see small red star representing phosphorylation of transcription factor bound to enhancer in the illustration).[72] An activated enhancer begins transcription of its RNA before activating transcription of messenger RNA from its target gene.[73]
DNA methylation and demethylation in transcriptional regulation
[edit]
DNA methylation is a widespread mechanism for epigenetic influence on gene expression and is seen in bacteria and eukaryotes and has roles in heritable transcription silencing and transcription regulation. Methylation most often occurs on a cytosine (see Figure). Methylation of cytosine primarily occurs in dinucleotide sequences where a cytosine is followed by a guanine, a CpG site. The number of CpG sites in the human genome is about 28 million.[74] Depending on the type of cell, about 70% of the CpG sites have a methylated cytosine.[75]
Methylation of cytosine in DNA has a major role in regulating gene expression. Methylation of CpGs in a promoter region of a gene usually represses gene transcription[76] while methylation of CpGs in the body of a gene increases expression.[77] TET enzymes play a central role in demethylation of methylated cytosines. Demethylation of CpGs in a gene promoter by TET enzyme activity increases transcription of the gene.[78]
Transcriptional regulation in learning and memory
[edit]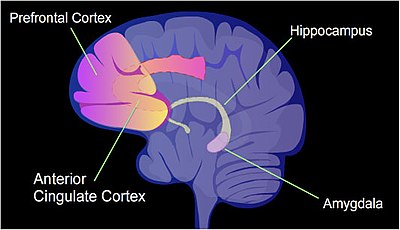
In a rat, contextual fear conditioning (CFC) is a painful learning experience. Just one episode of CFC can result in a life-long fearful memory.[79] After an episode of CFC, cytosine methylation is altered in the promoter regions of about 9.17% of all genes in the hippocampus neuron DNA of a rat.[80] The hippocampus is where new memories are initially stored. After CFC about 500 genes have increased transcription (often due to demethylation of CpG sites in a promoter region) and about 1,000 genes have decreased transcription (often due to newly formed 5-methylcytosine at CpG sites in a promoter region). The pattern of induced and repressed genes within neurons appears to provide a molecular basis for forming the first transient memory of this training event in the hippocampus of the rat brain.[80]
Some specific mechanisms guiding new DNA methylations and new DNA demethylations in the hippocampus during memory establishment have been established (see[81] for summary). One mechanism includes guiding the short isoform of the TET1 DNA demethylation enzyme, TET1s, to about 600 locations on the genome. The guidance is performed by association of TET1s with EGR1 protein, a transcription factor important in memory formation. Bringing TET1s to these locations initiates DNA demethylation at those sites, up-regulating associated genes. A second mechanism involves DNMT3A2, a splice-isoform of DNA methyltransferase DNMT3A, which adds methyl groups to cytosines in DNA. This isoform is induced by synaptic activity, and its location of action appears to be determined by histone post-translational modifications (a histone code). The resulting new messenger RNAs are then transported by messenger RNP particles (neuronal granules) to synapses of the neurons, where they can be translated into proteins affecting the activities of synapses.[81]
In particular, the brain-derived neurotrophic factor gene (BDNF) is known as a "learning gene".[82] After CFC there was upregulation of BDNF gene expression, related to decreased CpG methylation of certain internal promoters of the gene, and this was correlated with learning.[82]
Transcriptional regulation in cancer
[edit]The majority of gene promoters contain a CpG island with numerous CpG sites.[83] When many of a gene's promoter CpG sites are methylated the gene becomes silenced.[84] Colorectal cancers typically have 3 to 6 driver mutations and 33 to 66 hitchhiker or passenger mutations.[85] However, transcriptional silencing may be of more importance than mutation in causing progression to cancer. For example, in colorectal cancers about 600 to 800 genes are transcriptionally silenced by CpG island methylation (see regulation of transcription in cancer). Transcriptional repression in cancer can also occur by other epigenetic mechanisms, such as altered expression of microRNAs.[86] In breast cancer, transcriptional repression of BRCA1 may occur more frequently by over-transcribed microRNA-182 than by hypermethylation of the BRCA1 promoter (see Low expression of BRCA1 in breast and ovarian cancers).
Post-transcriptional regulation
[edit]In eukaryotes, where export of RNA is required before translation is possible, nuclear export is thought to provide additional control over gene expression. All transport in and out of the nucleus is via the nuclear pore and transport is controlled by a wide range of importin and exportin proteins.[87]
Expression of a gene coding for a protein is only possible if the messenger RNA carrying the code survives long enough to be translated.[41] In a typical cell, an RNA molecule is only stable if specifically protected from degradation.[88] RNA degradation has particular importance in regulation of expression in eukaryotic cells where mRNA has to travel significant distances before being translated.[89] In eukaryotes, RNA is stabilised by certain post-transcriptional modifications, particularly the 5′ cap and poly-adenylated tail.[90]
Intentional degradation of mRNA is used not just as a defence mechanism from foreign RNA (normally from viruses) but also as a route of mRNA destabilisation.[91] If an mRNA molecule has a complementary sequence to a small interfering RNA then it is targeted for destruction via the RNA interference pathway.[92]
Three prime untranslated regions and microRNAs
[edit]Three prime untranslated regions (3′UTRs) of messenger RNAs (mRNAs) often contain regulatory sequences that post-transcriptionally influence gene expression. Such 3′-UTRs often contain both binding sites for microRNAs (miRNAs) as well as for regulatory proteins.[93] By binding to specific sites within the 3′-UTR, miRNAs can decrease gene expression of various mRNAs by either inhibiting translation or directly causing degradation of the transcript.[94] The 3′-UTR also may have silencer regions that bind repressor proteins that inhibit the expression of a mRNA.[95]
The 3′-UTR often contains microRNA response elements (MREs). MREs are sequences to which miRNAs bind. These are prevalent motifs within 3′-UTRs. Among all regulatory motifs within the 3′-UTRs (e.g. including silencer regions), MREs make up about half of the motifs.[96]
As of 2014, the miRBase web site,[97] an archive of miRNA sequences and annotations, listed 28,645 entries in 233 biologic species. Of these, 1,881 miRNAs were in annotated human miRNA loci. miRNAs were predicted to have an average of about four hundred target mRNAs (affecting expression of several hundred genes).[98] Friedman et al.[98] estimate that >45,000 miRNA target sites within human mRNA 3′UTRs are conserved above background levels, and >60% of human protein-coding genes have been under selective pressure to maintain pairing to miRNAs.
Direct experiments show that a single miRNA can reduce the stability of hundreds of unique mRNAs.[99] Other experiments show that a single miRNA may repress the production of hundreds of proteins, but that this repression often is relatively mild (less than 2-fold).[100][101]
The effects of miRNA dysregulation of gene expression seem to be important in cancer.[102] For instance, in gastrointestinal cancers, nine miRNAs have been identified as epigenetically altered and effective in down regulating DNA repair enzymes.[103]
The effects of miRNA dysregulation of gene expression also seem to be important in neuropsychiatric disorders, such as schizophrenia, bipolar disorder, major depression, Parkinson's disease, Alzheimer's disease and autism spectrum disorders.[104][105]
Translational regulation
[edit]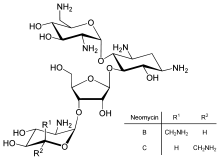
Direct regulation of translation is less prevalent than control of transcription or mRNA stability but is occasionally used.[106] Inhibition of protein translation is a major target for toxins and antibiotics, so they can kill a cell by overriding its normal gene expression control.[107] Protein synthesis inhibitors include the antibiotic neomycin and the toxin ricin.[108]
Post-translational modifications
[edit]Post-translational modifications (PTMs) are covalent modifications to proteins. Like RNA splicing, they help to significantly diversify the proteome. These modifications are usually catalyzed by enzymes. Additionally, processes like covalent additions to amino acid side chain residues can often be reversed by other enzymes. However, some, like the proteolytic cleavage of the protein backbone, are irreversible.[109]
PTMs play many important roles in the cell.[110] For example, phosphorylation is primarily involved in activating and deactivating proteins and in signaling pathways.[111] PTMs are involved in transcriptional regulation: an important function of acetylation and methylation is histone tail modification, which alters how accessible DNA is for transcription.[109] They can also be seen in the immune system, where glycosylation plays a key role.[112] One type of PTM can initiate another type of PTM, as can be seen in how ubiquitination tags proteins for degradation through proteolysis.[109] Proteolysis, other than being involved in breaking down proteins, is also important in activating and deactivating them, and in regulating biological processes such as DNA transcription and cell death.[113]
Measurement
[edit]
Measuring gene expression is an important part of many life sciences, as the ability to quantify the level at which a particular gene is expressed within a cell, tissue or organism can provide a lot of valuable information. For example, measuring gene expression can:
- Identify viral infection of a cell (viral protein expression).
- Determine an individual's susceptibility to cancer (oncogene expression).
- Find if a bacterium is resistant to penicillin (beta-lactamase expression).
- Gene expression profiling evaluates a panel of genes to help understand the fundamental mechanism of a cell. This is increasingly used in cancer therapy to target specific chemotherapy. (See RNA-Seq and DNA_microarray for details.)
Similarly, the analysis of the location of protein expression is a powerful tool, and this can be done on an organismal or cellular scale. Investigation of localization is particularly important for the study of development in multicellular organisms and as an indicator of protein function in single cells. Ideally, measurement of expression is done by detecting the final gene product (for many genes, this is the protein); however, it is often easier to detect one of the precursors, typically mRNA and to infer gene-expression levels from these measurements.
mRNA quantification
[edit]Levels of mRNA can be quantitatively measured by northern blotting, which provides size and sequence information about the mRNA molecules.[114] A sample of RNA is separated on an agarose gel and hybridized to a radioactively labeled RNA probe that is complementary to the target sequence.[115] The radiolabeled RNA is then detected by an autoradiograph.[116] Because the use of radioactive reagents makes the procedure time-consuming and potentially dangerous, alternative labeling and detection methods, such as digoxigenin and biotin chemistries, have been developed.[117] Perceived disadvantages of Northern blotting are that large quantities of RNA are required and that quantification may not be completely accurate, as it involves measuring band strength in an image of a gel.[118] On the other hand, the additional mRNA size information from the Northern blot allows the discrimination of alternately spliced transcripts.[119][120]
Another approach for measuring mRNA abundance is RT-qPCR. In this technique, reverse transcription is followed by quantitative PCR. Reverse transcription first generates a DNA template from the mRNA; this single-stranded template is called cDNA. The cDNA template is then amplified in the quantitative step, during which the fluorescence emitted by labeled hybridization probes or intercalating dyes changes as the DNA amplification process progresses.[121] With a carefully constructed standard curve, qPCR can produce an absolute measurement of the number of copies of original mRNA, typically in units of copies per nanolitre of homogenized tissue or copies per cell.[122] qPCR is very sensitive (detection of a single mRNA molecule is theoretically possible), but can be expensive depending on the type of reporter used; fluorescently labeled oligonucleotide probes are more expensive than non-specific intercalating fluorescent dyes.[123]
For expression profiling, or high-throughput analysis of many genes within a sample, quantitative PCR may be performed for hundreds of genes simultaneously in the case of low-density arrays.[124] A second approach is the hybridization microarray. A single array or "chip" may contain probes to determine transcript levels for every known gene in the genome of one or more organisms.[125] Alternatively, "tag based" technologies like Serial analysis of gene expression (SAGE) and RNA-Seq, which can provide a relative measure of the cellular concentration of different mRNAs, can be used.[126] An advantage of tag-based methods is the "open architecture", allowing for the exact measurement of any transcript, with a known or unknown sequence.[127] Next-generation sequencing (NGS) such as RNA-Seq is another approach, producing vast quantities of sequence data that can be matched to a reference genome. Although NGS is comparatively time-consuming, expensive, and resource-intensive, it can identify single-nucleotide polymorphisms, splice-variants, and novel genes, and can also be used to profile expression in organisms for which little or no sequence information is available.[128]
RNA profiles in Wikipedia
[edit]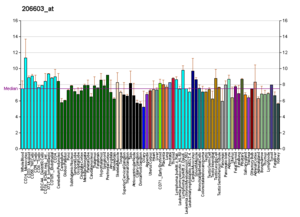
Profiles like these are found for almost all proteins listed in Wikipedia. They are generated by organizations such as the Genomics Institute of the Novartis Research Foundation and the European Bioinformatics Institute. Additional information can be found by searching their databases (for an example of the GLUT4 transporter pictured here, see citation).[129] These profiles indicate the level of DNA expression (and hence RNA produced) of a certain protein in a certain tissue, and are color-coded accordingly in the images located in the Protein Box on the right side of each Wikipedia page.
Protein quantification
[edit]For genes encoding proteins, the expression level can be directly assessed by a number of methods with some clear analogies to the techniques for mRNA quantification.
One of the most commonly used methods is to perform a Western blot against the protein of interest.[130] This gives information on the size of the protein in addition to its identity. A sample (often cellular lysate) is separated on a polyacrylamide gel, transferred to a membrane and then probed with an antibody to the protein of interest. The antibody can either be conjugated to a fluorophore or to horseradish peroxidase for imaging and/or quantification. The gel-based nature of this assay makes quantification less accurate, but it has the advantage of being able to identify later modifications to the protein, for example proteolysis or ubiquitination, from changes in size.
mRNA-protein correlation
[edit]While transcription directly reflects gene expression, the copy number of mRNA molecules does not directly correlate with the number of protein molecules translated from mRNA. Quantification of both protein and mRNA permits a correlation of the two levels. Regulation on each step of gene expression can impact the correlation, as shown for regulation of translation[28] or protein stability.[131] Post-translational factors, such as protein transport in highly polar cells,[132] can influence the measured mRNA-protein correlation as well.
Localization
[edit]
Analysis of expression is not limited to quantification; localization can also be determined. mRNA can be detected with a suitably labelled complementary mRNA strand and protein can be detected via labelled antibodies. The probed sample is then observed by microscopy to identify where the mRNA or protein is.
By replacing the gene with a new version fused to a green fluorescent protein marker or similar, expression may be directly quantified in live cells. This is done by imaging using a fluorescence microscope. It is very difficult to clone a GFP-fused protein into its native location in the genome without affecting expression levels, so this method often cannot be used to measure endogenous gene expression. It is, however, widely used to measure the expression of a gene artificially introduced into the cell, for example via an expression vector. By fusing a target protein to a fluorescent reporter, the protein's behavior, including its cellular localization and expression level, can be significantly changed.
The enzyme-linked immunosorbent assay works by using antibodies immobilised on a microtiter plate to capture proteins of interest from samples added to the well. Using a detection antibody conjugated to an enzyme or fluorophore the quantity of bound protein can be accurately measured by fluorometric or colourimetric detection. The detection process is very similar to that of a Western blot, but by avoiding the gel steps more accurate quantification can be achieved.
Expression system
[edit]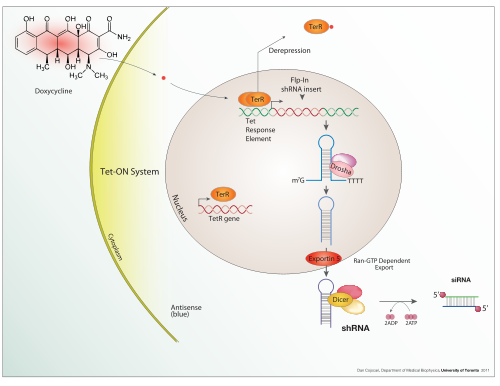
An expression system is a system specifically designed for the production of a gene product of choice. This is normally a protein although may also be RNA, such as tRNA or a ribozyme. An expression system consists of a gene, normally encoded by DNA, and the molecular machinery required to transcribe the DNA into mRNA and translate the mRNA into protein using the reagents provided. In the broadest sense this includes every living cell but the term is more normally used to refer to expression as a laboratory tool. An expression system is therefore often artificial in some manner. Expression systems are, however, a fundamentally natural process. Viruses are an excellent example where they replicate by using the host cell as an expression system for the viral proteins and genome.
Inducible expression
[edit]Doxycycline is also used in "Tet-on" and "Tet-off" tetracycline controlled transcriptional activation to regulate transgene expression in organisms and cell cultures.
In nature
[edit]In addition to these biological tools, certain naturally observed configurations of DNA (genes, promoters, enhancers, repressors) and the associated machinery itself are referred to as an expression system. This term is normally used in the case where a gene or set of genes is switched on under well defined conditions, for example, the simple repressor switch expression system in Lambda phage and the lac operator system in bacteria. Several natural expression systems are directly used or modified and used for artificial expression systems such as the Tet-on and Tet-off expression system.
Gene networks
[edit]Genes have sometimes been regarded as nodes in a network, with inputs being proteins such as transcription factors, and outputs being the level of gene expression. The node itself performs a function, and the operation of these functions have been interpreted as performing a kind of information processing within cells and determines cellular behavior.
Gene networks can also be constructed without formulating an explicit causal model. This is often the case when assembling networks from large expression data sets.[133] Covariation and correlation of expression is computed across a large sample of cases and measurements (often transcriptome or proteome data). The source of variation can be either experimental or natural (observational). There are several ways to construct gene expression networks, but one common approach is to compute a matrix of all pair-wise correlations of expression across conditions, time points, or individuals and convert the matrix (after thresholding at some cut-off value) into a graphical representation in which nodes represent genes, transcripts, or proteins and edges connecting these nodes represent the strength of association (see GeneNetwork GeneNetwork 2).[134]
Techniques and tools
[edit]The following experimental techniques are used to measure gene expression and are listed in roughly chronological order, starting with the older, more established technologies. They are divided into two groups based on their degree of multiplexity.
- Low-to-mid-plex techniques:
- Higher-plex techniques:
Gene expression databases
[edit]- Gene expression omnibus (GEO) at NCBI[140]
- Expression Atlas at the EBI
- Bgee Bgee at the SIB Swiss Institute of Bioinformatics
- Mouse Gene Expression Database at the Jackson Laboratory
- CollecTF: a database of experimentally validated transcription factor-binding sites in Bacteria.[141]
- COLOMBOS: collection of bacterial expression compendia.[142]
- Many Microbe Microarrays Database: microbial Affymetrix data[143]
See also
[edit]- AlloMap molecular expression testing
- Bookmarking
- EPIC-Seq
- Expressed sequence tag
- Expression Atlas
- Expression profiling
- Gene structure
- Genetic engineering
- Genetically modified organism
- List of biological databases
- List of human genes
- Oscillating gene
- Paramutation
- Protein production
- Protein purification
- Ribonomics
- Ridge
- Sequence profiling tool
- Transcriptional bursting
- Transcriptional noise
- Transcript of unknown function
References
[edit]- ^ Brueckner F, Armache KJ, Cheung A, Damsma GE, Kettenberger H, Lehmann E, et al. (February 2009). "Structure-function studies of the RNA polymerase II elongation complex". Acta Crystallographica. Section D, Biological Crystallography. 65 (Pt 2): 112–120. Bibcode:2009AcCrD..65..112B. doi:10.1107/S0907444908039875. PMC 2631633. PMID 19171965.
- ^ Krebs JE, Goldstein ES, Kilpatrick ST (2017-03-02). Lewin's genes XII. Burlington, MA: Jones & Bartlett Learning. ISBN 978-1-284-10449-3. OCLC 965781334.
- ^ Ramanathan A, Robb GB, Chan SH (September 2016). "mRNA capping: biological functions and applications". Nucleic Acids Research. 44 (16): 7511–7526. doi:10.1093/nar/gkw551. PMC 5027499. PMID 27317694.
- ^ Gonatopoulos-Pournatzis T, Cowling VH (January 2014). "Cap-binding complex (CBC)". The Biochemical Journal. 457 (2): 231–242. doi:10.1042/BJ20131214. PMC 3901397. PMID 24354960.
- ^ Neve J, Patel R, Wang Z, Louey A, Furger AM (July 2017). "Cleavage and polyadenylation: Ending the message expands gene regulation". RNA Biology. 14 (7): 865–890. doi:10.1080/15476286.2017.1306171. PMC 5546720. PMID 28453393.
- ^ Borodulina OR, Kramerov DA (September 2008). "Transcripts synthesized by RNA polymerase III can be polyadenylated in an AAUAAA-dependent manner". RNA. 14 (9): 1865–1873. doi:10.1261/rna.1006608. PMC 2525947. PMID 18658125.
- ^ Munoz-Tello P, Rajappa L, Coquille S, Thore S (2015). "Polyuridylation in Eukaryotes: A 3'-End Modification Regulating RNA Life". BioMed Research International. 2015: 968127. doi:10.1155/2015/968127. PMC 4442281. PMID 26078976.
- ^ Passmore LA, Coller J (February 2022). "Roles of mRNA poly(A) tails in regulation of eukaryotic gene expression". Nature Reviews. Molecular Cell Biology. 23 (2): 93–106. doi:10.1038/s41580-021-00417-y. PMC 7614307. PMID 34594027.
- ^ Morozov IY, Jones MG, Razak AA, Rigden DJ, Caddick MX (January 2010). "CUCU modification of mRNA promotes decapping and transcript degradation in Aspergillus nidulans". Molecular and Cellular Biology. 30 (2): 460–469. doi:10.1128/MCB.00997-09. PMC 2798463. PMID 19901075.
- ^ Darnell JE (April 2013). "Reflections on the history of pre-mRNA processing and highlights of current knowledge: a unified picture". RNA. 19 (4): 443–460. doi:10.1261/rna.038596.113. PMC 3677254. PMID 23440351.
- ^ Zhang L, Vielle A, Espinosa S, Zhao R (May 2019). "RNAs in the spliceosome: Insight from cryoEM structures". Wiley Interdisciplinary Reviews. RNA. 10 (3): e1523. doi:10.1002/wrna.1523. PMC 6450755. PMID 30729694.
- ^ Hossain MA, Rodriguez CM, Johnson TL (October 2011). "Key features of the two-intron Saccharomyces cerevisiae gene SUS1 contribute to its alternative splicing". Nucleic Acids Research. 39 (19): 8612–8627. doi:10.1093/nar/gkr497. PMC 3201863. PMID 21749978.
- ^ Baralle FE, Giudice J (July 2017). "Alternative splicing as a regulator of development and tissue identity". Nature Reviews. Molecular Cell Biology. 18 (7): 437–451. doi:10.1038/nrm.2017.27. PMC 6839889. PMID 28488700.
- ^ Baum B, Spang A (December 2023). "On the origin of the nucleus: a hypothesis". Microbiology and Molecular Biology Reviews. 87 (4): e0018621. doi:10.1128/mmbr.00186-21. PMC 10732040. PMID 38018971.
- ^ Sirri V, Urcuqui-Inchima S, Roussel P, Hernandez-Verdun D (January 2008). "Nucleolus: the fascinating nuclear body". Histochemistry and Cell Biology. 129 (1): 13–31. doi:10.1007/s00418-007-0359-6. PMC 2137947. PMID 18046571.
- ^ Frank DN, Pace NR (1998). "Ribonuclease P: unity and diversity in a tRNA processing ribozyme". Annual Review of Biochemistry. 67: 153–180. doi:10.1146/annurev.biochem.67.1.153. PMID 9759486.
- ^ Ceballos M, Vioque A (2007). "tRNase Z". Protein and Peptide Letters. 14 (2): 137–145. doi:10.2174/092986607779816050. PMID 17305600.
- ^ Weiner AM (October 2004). "tRNA maturation: RNA polymerization without a nucleic acid template". Current Biology. 14 (20): R883–R885. Bibcode:2004CBio...14.R883W. doi:10.1016/j.cub.2004.09.069. PMID 15498478.
- ^ Bratkovič T, Božič J, Rogelj B (February 2020). "Functional diversity of small nucleolar RNAs". Nucleic Acids Research. 48 (4): 1627–1651. doi:10.1093/nar/gkz1140. PMC 7038934. PMID 31828325.
- ^ Nizami Z, Deryusheva S, Gall JG (July 2010). "The Cajal body and histone locus body". Cold Spring Harbor Perspectives in Biology. 2 (7): a000653. doi:10.1101/cshperspect.a000653. PMC 2890199. PMID 20504965.
- ^ Darzacq X, Jády BE, Verheggen C, Kiss AM, Bertrand E, Kiss T (June 2002). "Cajal body-specific small nuclear RNAs: a novel class of 2'-O-methylation and pseudouridylation guide RNAs". The EMBO Journal. 21 (11): 2746–2756. doi:10.1093/emboj/21.11.2746. PMC 126017. PMID 12032087.
- ^ Köhler A, Hurt E (October 2007). "Exporting RNA from the nucleus to the cytoplasm". Nature Reviews. Molecular Cell Biology. 8 (10): 761–773. doi:10.1038/nrm2255. PMID 17786152. S2CID 10836137.
- ^ Jambhekar A, Derisi JL (May 2007). "Cis-acting determinants of asymmetric, cytoplasmic RNA transport". RNA. 13 (5): 625–642. doi:10.1261/rna.262607. PMC 1852811. PMID 17449729.
- ^ Amaral PP, Dinger ME, Mercer TR, Mattick JS (March 2008). "The eukaryotic genome as an RNA machine". Science. 319 (5871): 1787–1789. Bibcode:2008Sci...319.1787A. doi:10.1126/science.1155472. PMID 18369136. S2CID 206511756.
- ^ Hansen TM, Baranov PV, Ivanov IP, Gesteland RF, Atkins JF (May 2003). "Maintenance of the correct open reading frame by the ribosome". EMBO Reports. 4 (5): 499–504. doi:10.1038/sj.embor.embor825. PMC 1319180. PMID 12717454.
- ^ Berk V, Cate JH (June 2007). "Insights into protein biosynthesis from structures of bacterial ribosomes". Current Opinion in Structural Biology. 17 (3): 302–309. doi:10.1016/j.sbi.2007.05.009. PMID 17574829.
- ^ Schwanhäusser B, Busse D, Li N, Dittmar G, Schuchhardt J, Wolf J, et al. (May 2011). "Global quantification of mammalian gene expression control" (PDF). Nature. 473 (7347): 337–342. Bibcode:2011Natur.473..337S. doi:10.1038/nature10098. PMID 21593866. S2CID 205224972.
- ^ a b Schwanhäusser B, Busse D, Li N, Dittmar G, Schuchhardt J, Wolf J, et al. (March 2013). "Corrigendum: Global quantification of mammalian gene expression control". Nature. 495 (7439): 126–127. Bibcode:2013Natur.495..126S. doi:10.1038/nature11848. PMID 23407496.
- ^ Hegde RS, Kang SW (July 2008). "The concept of translocational regulation". The Journal of Cell Biology. 182 (2): 225–232. doi:10.1083/jcb.200804157. PMC 2483521. PMID 18644895.
- ^ Alberts B, Johnson A, Lewis J, Raff M, Roberts K, Walters P (2002). "The Shape and Structure of Proteins". Molecular Biology of the Cell (Fourth ed.). New York and London: Garland Science. ISBN 978-0-8153-3218-3.
- ^ Anfinsen CB (July 1972). "The formation and stabilization of protein structure". The Biochemical Journal. 128 (4): 737–749. doi:10.1042/bj1280737. PMC 1173893. PMID 4565129.
- ^ Berg JM, Tymoczko JL, Lubert Stryer; Web content by Neil D. Clarke (2002). "3. Protein Structure and Function". Biochemistry. San Francisco: W. H. Freeman. ISBN 978-0-7167-4684-3.
- ^ Selkoe DJ (December 2003). "Folding proteins in fatal ways". Nature. 426 (6968): 900–904. Bibcode:2003Natur.426..900S. doi:10.1038/nature02264. PMID 14685251. S2CID 6451881.
- ^ Alberts B, Bray D, Hopkin K, Johnson A, Lewis J, Raff M, et al. (2010). "Protein Structure and Function". Essential Cell Biology (3rd ed.). New York: Garland Science, Taylor and Francis Group, LLC. pp. 120–170. ISBN 978-0-8153-4129-1.
- ^ Hebert DN, Molinari M (October 2007). "In and out of the ER: protein folding, quality control, degradation, and related human diseases". Physiological Reviews. 87 (4): 1377–1408. doi:10.1152/physrev.00050.2006. PMID 17928587.
- ^ Russell R (January 2008). "RNA misfolding and the action of chaperones". Frontiers in Bioscience. 13 (13): 1–20. doi:10.2741/2557. PMC 2610265. PMID 17981525.
- ^ Kober L, Zehe C, Bode J (April 2013). "Optimized signal peptides for the development of high expressing CHO cell lines". Biotechnology and Bioengineering. 110 (4): 1164–1173. doi:10.1002/bit.24776. PMID 23124363. S2CID 449870.
- ^ Lu J, Wu T, Zhang B, Liu S, Song W, Qiao J, et al. (May 2021). "Types of nuclear localization signals and mechanisms of protein import into the nucleus". Cell Communication and Signaling. 19 (1): 60. doi:10.1186/s12964-021-00741-y. PMC 8140498. PMID 34022911.
- ^ Lang S, Nguyen D, Bhadra P, Jung M, Helms V, Zimmermann R (2022-07-11). "Signal Peptide Features Determining the Substrate Specificities of Targeting and Translocation Components in Human ER Protein Import". Frontiers in Physiology. 13: 833540. doi:10.3389/fphys.2022.833540. PMC 9309488. PMID 35899032.
- ^ Murat D, Byrne M, Komeili A (October 2010). "Cell biology of prokaryotic organelles". Cold Spring Harbor Perspectives in Biology. 2 (10): a000422. doi:10.1101/cshperspect.a000422. PMC 2944366. PMID 20739411.
- ^ a b Alberts B, Johnson A, Lewis J, Raff M, Roberts K, Walter P (2002), "From RNA to Protein", Molecular Biology of the Cell. 4th edition, Garland Science, retrieved 2024-06-10
- ^ Moreau P, Brandizzi F, Hanton S, Chatre L, Melser S, Hawes C, et al. (2007). "The plant ER-Golgi interface: a highly structured and dynamic membrane complex". Journal of Experimental Botany. 58 (1): 49–64. doi:10.1093/jxb/erl135. PMID 16990376.
- ^ Prudovsky I, Tarantini F, Landriscina M, Neivandt D, Soldi R, Kirov A, et al. (April 2008). "Secretion without Golgi". Journal of Cellular Biochemistry. 103 (5): 1327–1343. doi:10.1002/jcb.21513. PMC 2613191. PMID 17786931.
- ^ Zaidi SK, Young DW, Choi JY, Pratap J, Javed A, Montecino M, et al. (October 2004). "Intranuclear trafficking: organization and assembly of regulatory machinery for combinatorial biological control". The Journal of Biological Chemistry. 279 (42): 43363–43366. doi:10.1074/jbc.R400020200. PMID 15277516.
- ^ Mattick JS, Amaral PP, Dinger ME, Mercer TR, Mehler MF (January 2009). "RNA regulation of epigenetic processes". BioEssays. 31 (1): 51–59. doi:10.1002/bies.080099. PMID 19154003.
- ^ Martinez NJ, Walhout AJ (April 2009). "The interplay between transcription factors and microRNAs in genome-scale regulatory networks". BioEssays. 31 (4): 435–445. doi:10.1002/bies.200800212. PMC 3118512. PMID 19274664.
- ^ Tomilin NV (April 2008). "Regulation of mammalian gene expression by retroelements and non-coding tandem repeats". BioEssays. 30 (4): 338–348. doi:10.1002/bies.20741. PMID 18348251.
- ^ Lee TI, Young RA (March 2013). "Transcriptional regulation and its misregulation in disease". Cell. 152 (6): 1237–1251. doi:10.1016/j.cell.2013.02.014. PMC 3640494. PMID 23498934.
- ^ O'Connor L, Gilmour J, Bonifer C (December 2016). "The Role of the Ubiquitously Expressed Transcription Factor Sp1 in Tissue-specific Transcriptional Regulation and in Disease". The Yale Journal of Biology and Medicine. 89 (4): 513–525. PMC 5168829. PMID 28018142.
- ^ Yesudhas D, Batool M, Anwar MA, Panneerselvam S, Choi S (August 2017). "Proteins Recognizing DNA: Structural Uniqueness and Versatility of DNA-Binding Domains in Stem Cell Transcription Factors". Genes. 8 (8): 192. doi:10.3390/genes8080192. PMC 5575656. PMID 28763006.
- ^ Wang G, Wang F, Huang Q, Li Y, Liu Y, Wang Y (2015). "Understanding Transcription Factor Regulation by Integrating Gene Expression and DNase I Hypersensitive Sites". BioMed Research International. 2015: 757530. doi:10.1155/2015/757530. PMC 4573618. PMID 26425553.
- ^ Kolovos P, Knoch TA, Grosveld FG, Cook PR, Papantonis A (January 2012). "Enhancers and silencers: an integrated and simple model for their function". Epigenetics & Chromatin. 5 (1): 1. doi:10.1186/1756-8935-5-1. PMC 3281776. PMID 22230046.
- ^ Fuda NJ, Ardehali MB, Lis JT (September 2009). "Defining mechanisms that regulate RNA polymerase II transcription in vivo". Nature. 461 (7261): 186–192. Bibcode:2009Natur.461..186F. doi:10.1038/nature08449. PMC 2833331. PMID 19741698.
- ^ Filtz TM, Vogel WK, Leid M (February 2014). "Regulation of transcription factor activity by interconnected post-translational modifications". Trends in Pharmacological Sciences. 35 (2): 76–85. doi:10.1016/j.tips.2013.11.005. PMC 3954851. PMID 24388790.
- ^ Hampsey M (June 1998). "Molecular genetics of the RNA polymerase II general transcriptional machinery". Microbiology and Molecular Biology Reviews. 62 (2): 465–503. doi:10.1128/MMBR.62.2.465-503.1998. PMC 98922. PMID 9618449.
- ^ Veitia RA (November 2008). "One thousand and one ways of making functionally similar transcriptional enhancers". BioEssays. 30 (11–12): 1052–1057. doi:10.1002/bies.20849. PMID 18937349.
- ^ Nguyen T, Nioi P, Pickett CB (May 2009). "The Nrf2-antioxidant response element signaling pathway and its activation by oxidative stress". The Journal of Biological Chemistry. 284 (20): 13291–13295. doi:10.1074/jbc.R900010200. PMC 2679427. PMID 19182219.
- ^ Paul S (November 2008). "Dysfunction of the ubiquitin-proteasome system in multiple disease conditions: therapeutic approaches". BioEssays. 30 (11–12): 1172–1184. doi:10.1002/bies.20852. PMID 18937370. S2CID 29422790.
- ^ Los M, Maddika S, Erb B, Schulze-Osthoff K (May 2009). "Switching Akt: from survival signaling to deadly response". BioEssays. 31 (5): 492–495. doi:10.1002/bies.200900005. PMC 2954189. PMID 19319914.
- ^ Afek A, Sela I, Musa-Lempel N, Lukatsky DB (November 2011). "Nonspecific transcription-factor-DNA binding influences nucleosome occupancy in yeast". Biophysical Journal. 101 (10): 2465–2475. arXiv:1111.4779. Bibcode:2011BpJ...101.2465A. doi:10.1016/j.bpj.2011.10.012. PMC 3218343. PMID 22098745.
- ^ Moosavi A, Motevalizadeh Ardekani A (November 2016). "Role of Epigenetics in Biology and Human Diseases". Iranian Biomedical Journal. 20 (5): 246–258. doi:10.22045/ibj.2016.01. PMC 5075137. PMID 27377127.
- ^ Al Aboud NM, Tupper C, Jialal I (2024). "Genetics, Epigenetic Mechanism". StatPearls. Treasure Island (FL): StatPearls Publishing. PMID 30422591. Retrieved 2024-06-12.
- ^ Miller JL, Grant PA (2013). "The Role of DNA Methylation and Histone Modifications in Transcriptional Regulation in Humans". Epigenetics: Development and Disease. Subcellular Biochemistry. Vol. 61. pp. 289–317. doi:10.1007/978-94-007-4525-4_13. ISBN 978-94-007-4524-7. PMC 6611551. PMID 23150256.
- ^ Verheul TC, van Hijfte L, Perenthaler E, Barakat TS (2020). "The Why of YY1: Mechanisms of Transcriptional Regulation by Yin Yang 1". Frontiers in Cell and Developmental Biology. 8: 592164. doi:10.3389/fcell.2020.592164. PMC 7554316. PMID 33102493.
- ^ Spitz F, Furlong EE (September 2012). "Transcription factors: from enhancer binding to developmental control". Nature Reviews. Genetics. 13 (9): 613–626. doi:10.1038/nrg3207. PMID 22868264. S2CID 205485256.
- ^ a b Schoenfelder S, Fraser P (August 2019). "Long-range enhancer-promoter contacts in gene expression control". Nature Reviews. Genetics. 20 (8): 437–455. doi:10.1038/s41576-019-0128-0. PMID 31086298. S2CID 152283312.
- ^ Weintraub AS, Li CH, Zamudio AV, Sigova AA, Hannett NM, Day DS, et al. (December 2017). "YY1 Is a Structural Regulator of Enhancer-Promoter Loops". Cell. 171 (7): 1573–1588.e28. doi:10.1016/j.cell.2017.11.008. PMC 5785279. PMID 29224777.
- ^ Lambert SA, Jolma A, Campitelli LF, Das PK, Yin Y, Albu M, et al. (February 2018). "The Human Transcription Factors". Cell. 172 (4): 650–665. doi:10.1016/j.cell.2018.01.029. PMID 29425488.
- ^ Grossman SR, Engreitz J, Ray JP, Nguyen TH, Hacohen N, Lander ES (July 2018). "Positional specificity of different transcription factor classes within enhancers". Proceedings of the National Academy of Sciences of the United States of America. 115 (30): E7222–E7230. Bibcode:2018PNAS..115E7222G. doi:10.1073/pnas.1804663115. PMC 6065035. PMID 29987030.
- ^ Allen BL, Taatjes DJ (March 2015). "The Mediator complex: a central integrator of transcription". Nature Reviews. Molecular Cell Biology. 16 (3): 155–166. doi:10.1038/nrm3951. PMC 4963239. PMID 25693131.
- ^ Mikhaylichenko O, Bondarenko V, Harnett D, Schor IE, Males M, Viales RR, et al. (January 2018). "The degree of enhancer or promoter activity is reflected by the levels and directionality of eRNA transcription". Genes & Development. 32 (1): 42–57. doi:10.1101/gad.308619.117. PMC 5828394. PMID 29378788.
- ^ Li QJ, Yang SH, Maeda Y, Sladek FM, Sharrocks AD, Martins-Green M (January 2003). "MAP kinase phosphorylation-dependent activation of Elk-1 leads to activation of the co-activator p300". The EMBO Journal. 22 (2): 281–291. doi:10.1093/emboj/cdg028. PMC 140103. PMID 12514134.
- ^ Carullo NV, Phillips Iii RA, Simon RC, Soto SA, Hinds JE, Salisbury AJ, et al. (September 2020). "Enhancer RNAs predict enhancer-gene regulatory links and are critical for enhancer function in neuronal systems". Nucleic Acids Research. 48 (17): 9550–9570. doi:10.1093/nar/gkaa671. PMC 7515708. PMID 32810208.
- ^ Lövkvist C, Dodd IB, Sneppen K, Haerter JO (June 2016). "DNA methylation in human epigenomes depends on local topology of CpG sites". Nucleic Acids Research. 44 (11): 5123–5132. doi:10.1093/nar/gkw124. PMC 4914085. PMID 26932361.
- ^ Jabbari K, Bernardi G (May 2004). "Cytosine methylation and CpG, TpG (CpA) and TpA frequencies". Gene. 333: 143–149. doi:10.1016/j.gene.2004.02.043. PMID 15177689.
- ^ Weber M, Hellmann I, Stadler MB, Ramos L, Pääbo S, Rebhan M, et al. (April 2007). "Distribution, silencing potential and evolutionary impact of promoter DNA methylation in the human genome". Nature Genetics. 39 (4): 457–466. doi:10.1038/ng1990. PMID 17334365. S2CID 22446734.
- ^ Yang X, Han H, De Carvalho DD, Lay FD, Jones PA, Liang G (October 2014). "Gene body methylation can alter gene expression and is a therapeutic target in cancer". Cancer Cell. 26 (4): 577–590. doi:10.1016/j.ccr.2014.07.028. PMC 4224113. PMID 25263941.
- ^ Maeder ML, Angstman JF, Richardson ME, Linder SJ, Cascio VM, Tsai SQ, et al. (December 2013). "Targeted DNA demethylation and activation of endogenous genes using programmable TALE-TET1 fusion proteins". Nature Biotechnology. 31 (12): 1137–1142. doi:10.1038/nbt.2726. PMC 3858462. PMID 24108092.
- ^ Kim JJ, Jung MW (2006). "Neural circuits and mechanisms involved in Pavlovian fear conditioning: a critical review". Neuroscience and Biobehavioral Reviews. 30 (2): 188–202. doi:10.1016/j.neubiorev.2005.06.005. PMC 4342048. PMID 16120461.
- ^ a b Duke CG, Kennedy AJ, Gavin CF, Day JJ, Sweatt JD (July 2017). "Experience-dependent epigenomic reorganization in the hippocampus". Learning & Memory. 24 (7): 278–288. doi:10.1101/lm.045112.117. PMC 5473107. PMID 28620075.
- ^ a b Bernstein C (2022). "DNA Methylation and Establishing Memory". Epigenetics Insights. 15: 25168657211072499. doi:10.1177/25168657211072499. PMC 8793415. PMID 35098021.
- ^ a b Keifer J (February 2017). "Primetime for Learning Genes". Genes. 8 (2): 69. doi:10.3390/genes8020069. PMC 5333058. PMID 28208656.
- ^ Saxonov S, Berg P, Brutlag DL (January 2006). "A genome-wide analysis of CpG dinucleotides in the human genome distinguishes two distinct classes of promoters". Proceedings of the National Academy of Sciences of the United States of America. 103 (5): 1412–1417. Bibcode:2006PNAS..103.1412S. doi:10.1073/pnas.0510310103. PMC 1345710. PMID 16432200.
- ^ Bird A (January 2002). "DNA methylation patterns and epigenetic memory". Genes & Development. 16 (1): 6–21. doi:10.1101/gad.947102. PMID 11782440.
- ^ Vogelstein B, Papadopoulos N, Velculescu VE, Zhou S, Diaz LA, Kinzler KW (March 2013). "Cancer genome landscapes". Science. 339 (6127): 1546–1558. Bibcode:2013Sci...339.1546V. doi:10.1126/science.1235122. PMC 3749880. PMID 23539594.
- ^ Tessitore A, Cicciarelli G, Del Vecchio F, Gaggiano A, Verzella D, Fischietti M, et al. (2014). "MicroRNAs in the DNA Damage/Repair Network and Cancer". International Journal of Genomics. 2014: 820248. doi:10.1155/2014/820248. PMC 3926391. PMID 24616890.
- ^ De Magistris P (October 2021). "The Great Escape: mRNA Export through the Nuclear Pore Complex". International Journal of Molecular Sciences. 22 (21): 11767. doi:10.3390/ijms222111767. PMC 8583845. PMID 34769195.
- ^ Liu H, Luo M, Wen JK (May 2014). "mRNA stability in the nucleus". Journal of Zhejiang University. Science. B. 15 (5): 444–454. doi:10.1631/jzus.B1400088. PMC 4076601. PMID 24793762.
- ^ Yan LL, Zaher HS (October 2019). "How do cells cope with RNA damage and its consequences?". The Journal of Biological Chemistry. 294 (41): 15158–15171. doi:10.1074/jbc.REV119.006513. PMC 6791314. PMID 31439666.
- ^ Passmore LA, Coller J (February 2022). "Roles of mRNA poly(A) tails in regulation of eukaryotic gene expression". Nature Reviews. Molecular Cell Biology. 23 (2): 93–106. doi:10.1038/s41580-021-00417-y. PMC 7614307. PMID 34594027.
- ^ Shehata SI, Watkins JM, Burke JM, Parker R (February 2024). "Mechanisms and consequences of mRNA destabilization during viral infections". Virology Journal. 21 (1): 38. doi:10.1186/s12985-024-02305-1. PMC 10848536. PMID 38321453.
- ^ Tang L, Chen HY, Hao NB, Tang B, Guo H, Yong X, et al. (October 2017). "microRNA inhibitors: Natural and artificial sequestration of microRNA". Cancer Letters. 407: 139–147. doi:10.1016/B978-0-444-64046-8.00282-2. ISBN 978-0-444-64047-5. PMC 7152241. PMID 28602827.
- ^ Mayr C (October 2019). "What Are 3' UTRs Doing?". Cold Spring Harbor Perspectives in Biology. 11 (10): a034728. doi:10.1101/cshperspect.a034728. PMC 6771366. PMID 30181377.
- ^ O'Brien J, Hayder H, Zayed Y, Peng C (2018-08-03). "Overview of MicroRNA Biogenesis, Mechanisms of Actions, and Circulation". Frontiers in Endocrinology. 9: 402. doi:10.3389/fendo.2018.00402. PMC 6085463. PMID 30123182.
- ^ Mayya VK, Duchaine TF (2019-01-24). "Ciphers and Executioners: How 3'-Untranslated Regions Determine the Fate of Messenger RNAs". Frontiers in Genetics. 10: 6. doi:10.3389/fgene.2019.00006. PMC 6357968. PMID 30740123.
- ^ Nair AA, Tang X, Thompson KJ, Vedell PT, Kalari KR, Subramanian S (June 2020). "Frequency of MicroRNA Response Elements Identifies Pathologically Relevant Signaling Pathways in Triple-Negative Breast Cancer". iScience. 23 (6): 101249. Bibcode:2020iSci...23j1249N. doi:10.1016/j.isci.2020.101249. PMC 7322352. PMID 32629614.
- ^ miRBase.org
- ^ a b Friedman RC, Farh KK, Burge CB, Bartel DP (January 2009). "Most mammalian mRNAs are conserved targets of microRNAs". Genome Research. 19 (1): 92–105. doi:10.1101/gr.082701.108. PMC 2612969. PMID 18955434.
- ^ Lim LP, Lau NC, Garrett-Engele P, Grimson A, Schelter JM, Castle J, et al. (February 2005). "Microarray analysis shows that some microRNAs downregulate large numbers of target mRNAs". Nature. 433 (7027): 769–773. Bibcode:2005Natur.433..769L. doi:10.1038/nature03315. PMID 15685193. S2CID 4430576.
- ^ Selbach M, Schwanhäusser B, Thierfelder N, Fang Z, Khanin R, Rajewsky N (September 2008). "Widespread changes in protein synthesis induced by microRNAs". Nature. 455 (7209): 58–63. Bibcode:2008Natur.455...58S. doi:10.1038/nature07228. PMID 18668040. S2CID 4429008.
- ^ Baek D, Villén J, Shin C, Camargo FD, Gygi SP, Bartel DP (September 2008). "The impact of microRNAs on protein output". Nature. 455 (7209): 64–71. Bibcode:2008Natur.455...64B. doi:10.1038/nature07242. PMC 2745094. PMID 18668037.
- ^ Palmero EI, de Campos SG, Campos M, de Souza NC, Guerreiro ID, Carvalho AL, et al. (July 2011). "Mechanisms and role of microRNA deregulation in cancer onset and progression". Genetics and Molecular Biology. 34 (3): 363–370. doi:10.1590/S1415-47572011000300001. PMC 3168173. PMID 21931505.
- ^ Bernstein C, Bernstein H (May 2015). "Epigenetic reduction of DNA repair in progression to gastrointestinal cancer". World Journal of Gastrointestinal Oncology. 7 (5): 30–46. doi:10.4251/wjgo.v7.i5.30. PMC 4434036. PMID 25987950.
- ^ Mellios N, Sur M (2012). "The Emerging Role of microRNAs in Schizophrenia and Autism Spectrum Disorders". Frontiers in Psychiatry. 3: 39. doi:10.3389/fpsyt.2012.00039. PMC 3336189. PMID 22539927.
- ^ Geaghan M, Cairns MJ (August 2015). "MicroRNA and Posttranscriptional Dysregulation in Psychiatry". Biological Psychiatry. 78 (4): 231–239. doi:10.1016/j.biopsych.2014.12.009. hdl:1959.13/1335073. PMID 25636176.
- ^ Sonenberg N, Hinnebusch AG (February 2009). "Regulation of translation initiation in eukaryotes: mechanisms and biological targets". Cell. 136 (4): 731–745. doi:10.1016/j.cell.2009.01.042. PMC 3610329. PMID 19239892.
- ^ Jurėnas D, Van Melderen L (2020-04-17). "The Variety in the Common Theme of Translation Inhibition by Type II Toxin-Antitoxin Systems". Frontiers in Genetics. 11: 262. doi:10.3389/fgene.2020.00262. PMC 7180214. PMID 32362907.
- ^ Dmitriev SE, Vladimirov DO, Lashkevich KA (November 2020). "A Quick Guide to Small-Molecule Inhibitors of Eukaryotic Protein Synthesis". Biochemistry. Biokhimiia. 85 (11): 1389–1421. doi:10.1134/S0006297920110097. PMC 7689648. PMID 33280581.
- ^ a b c Walsh CT, Garneau-Tsodikova S, Gatto GJ (December 2005). "Protein posttranslational modifications: the chemistry of proteome diversifications". Angewandte Chemie. 44 (45): 7342–7372. doi:10.1002/anie.200501023. PMID 16267872. S2CID 32157563.
- ^ Khoury GA, Baliban RC, Floudas CA (September 2011). "Proteome-wide post-translational modification statistics: frequency analysis and curation of the swiss-prot database". Scientific Reports. 1 (90): 90. Bibcode:2011NatSR...1...90K. doi:10.1038/srep00090. PMC 3201773. PMID 22034591.
- ^ Mann M, Jensen ON (March 2003). "Proteomic analysis of post-translational modifications". Nature Biotechnology. 21 (3): 255–261. doi:10.1038/nbt0303-255. PMID 12610572. S2CID 205266061.
- ^ Seo J, Lee KJ (January 2004). "Post-translational modifications and their biological functions: proteomic analysis and systematic approaches". Journal of Biochemistry and Molecular Biology. 37 (1): 35–44. doi:10.5483/bmbrep.2004.37.1.035. PMID 14761301.
- ^ Rogers LD, Overall CM (December 2013). "Proteolytic post-translational modification of proteins: proteomic tools and methodology". Molecular & Cellular Proteomics. 12 (12): 3532–3542. doi:10.1074/mcp.M113.031310. PMC 3861706. PMID 23887885.
- ^ Ahmad W, Gull B, Baby J, Mustafa F (June 2021). "A Comprehensive Analysis of Northern versus Liquid Hybridization Assays for mRNAs, Small RNAs, and miRNAs Using a Non-Radiolabeled Approach". Current Issues in Molecular Biology. 43 (2): 457–484. doi:10.3390/cimb43020036. PMC 8929067. PMID 34206608.
- ^ Hoy MA (2013). "Chapter 6 - Some Additional Tools for the Molecular Biologist". Insect Molecular Genetics (3rd ed.). Academic Press. pp. 215–249. doi:10.1016/C2011-0-69839-4. ISBN 978-0-12-415874-0.
- ^ Petrov A, Tsa A, Puglisi JD (2013). "Analysis of RNA by analytical polyacrylamide gel electrophoresis". Laboratory Methods in Enzymology: RNA. Vol. 530. pp. 301–313. doi:10.1016/B978-0-12-420037-1.00016-6. ISBN 978-0-12-420037-1. ISSN 1557-7988. PMID 24034328.
- ^ Boyle A, Perry-O'Keefe H (May 2001). "Labeling and colorimetric detection of nonisotopic probes". Current Protocols in Molecular Biology. 3 (Supplement 20): Unit3.18. doi:10.1002/0471142727.mb0318s20. PMID 18265226.
- ^ Kuang J, Yan X, Genders AJ, Granata C, Bishop DJ (2018-05-10). "An overview of technical considerations when using quantitative real-time PCR analysis of gene expression in human exercise research". PLOS ONE. 13 (5): e0196438. Bibcode:2018PLoSO..1396438K. doi:10.1371/journal.pone.0196438. PMC 5944930. PMID 29746477.
- ^ Yang T, Zhang M, Zhang N (January 2022). "Modified Northern blot protocol for easy detection of mRNAs in total RNA using radiolabeled probes". BMC Genomics. 23 (1): 66. doi:10.1186/s12864-021-08275-w. PMC 8772191. PMID 35057752.
- ^ Wesierska-Gadek J, Wang ZQ, Schmid G (January 1999). "Reduced stability of regularly spliced but not alternatively spliced p53 protein in PARP-deficient mouse fibroblasts". Cancer Research. 59 (1): 28–34. PMID 9892179.
- ^ Padhi BK, Singh M, Rosales M, Pelletier G, Cakmak S (May 2018). "A PCR-based quantitative assay for the evaluation of mRNA integrity in rat samples". Biomolecular Detection and Quantification. 15: 18–23. doi:10.1016/j.bdq.2018.02.001. PMC 6006387. PMID 29922590.
- ^ Kwon YM, Ricke SC (2011-03-22). High-Throughput Next Generation Sequencing. Methods in Molecular Biology. Vol. 733 (1st ed.). Springer Science+Business Media. doi:10.1007/978-1-61779-089-8. eISSN 1940-6029. ISBN 978-1-61779-089-8. ISSN 1064-3745.
- ^ Rahmasari R, Raekiansyah M, Azallea SN, Nethania M, Bilqisthy N, Rozaliyani A, et al. (November 2022). "Low-cost SYBR Green-based RT-qPCR assay for detecting SARS-CoV-2 in an Indonesian setting using WHO-recommended primers". Heliyon. 8 (11): e11130. Bibcode:2022Heliy...811130R. doi:10.1016/j.heliyon.2022.e11130. PMC 9617658. PMID 36339747.
- ^ Liang M, Cowley AW, Greene AS (January 2004). "High throughput gene expression profiling: a molecular approach to integrative physiology". The Journal of Physiology. 554 (Pt 1): 22–30. doi:10.1113/jphysiol.2003.049395. PMC 1664740. PMID 14678487.
- ^ Bumgarner R (January 2013). "Overview of DNA microarrays: types, applications, and their future". Current Protocols in Molecular Biology. Chapter 22: Unit 22.1. doi:10.1002/0471142727.mb2201s101. PMC 4011503. PMID 23288464.
- ^ Byron SA, Van Keuren-Jensen KR, Engelthaler DM, Carpten JD, Craig DW (May 2016). "Translating RNA sequencing into clinical diagnostics: opportunities and challenges". Nature Reviews. Genetics. 17 (5): 257–271. doi:10.1038/nrg.2016.10. PMC 7097555. PMID 26996076.
- ^ Meyers BC, Galbraith DW, Nelson T, Agrawal V (June 2004). "Methods for transcriptional profiling in plants. Be fruitful and replicate". Plant Physiology. 135 (2): 637–652. doi:10.1104/pp.104.040840. PMC 514100. PMID 15173570.
- ^ Kukurba KR, Montgomery SB (April 2015). "RNA Sequencing and Analysis". Cold Spring Harbor Protocols. 2015 (11): 951–969. doi:10.1101/pdb.top084970. PMC 4863231. PMID 25870306.
- ^ "Search results < Expression Atlas < EMBL-EBI". www.ebi.ac.uk.
- ^ Moritz CP (February 2020). "40 years Western blotting: A scientific birthday toast". Journal of Proteomics. 212: 103575. doi:10.1016/j.jprot.2019.103575. PMID 31706026.
- ^ Burkhart JM, Vaudel M, Gambaryan S, Radau S, Walter U, Martens L, et al. (October 2012). "The first comprehensive and quantitative analysis of human platelet protein composition allows the comparative analysis of structural and functional pathways". Blood. 120 (15): e73–e82. doi:10.1182/blood-2012-04-416594. PMID 22869793.
- ^ Moritz CP, Mühlhaus T, Tenzer S, Schulenborg T, Friauf E (June 2019). "Poor transcript-protein correlation in the brain: negatively correlating gene products reveal neuronal polarity as a potential cause". Journal of Neurochemistry. 149 (5): 582–604. doi:10.1111/jnc.14664. PMID 30664243. S2CID 58667771.
- ^ Banf M, Rhee SY (January 2017). "Computational inference of gene regulatory networks: Approaches, limitations and opportunities". Biochimica et Biophysica Acta (BBA) - Gene Regulatory Mechanisms. 1860 (1): 41–52. doi:10.1016/j.bbagrm.2016.09.003. PMID 27641093.
- ^ Chesler EJ, Lu L, Wang J, Williams RW, Manly KF (May 2004). "WebQTL: rapid exploratory analysis of gene expression and genetic networks for brain and behavior". Nature Neuroscience. 7 (5): 485–486. doi:10.1038/nn0504-485. PMID 15114364. S2CID 20241963.
- ^ Song Y, Wang W, Qu X, Sun S (February 2009). "Effects of hypoxia inducible factor-1alpha (HIF-1alpha) on the growth & adhesion in tongue squamous cell carcinoma cells". The Indian Journal of Medical Research. 129 (2): 154–163. PMID 19293442.
- ^ Hanriot L, Keime C, Gay N, Faure C, Dossat C, Wincker P, et al. (September 2008). "A combination of LongSAGE with Solexa sequencing is well suited to explore the depth and the complexity of transcriptome". BMC Genomics. 9: 418. doi:10.1186/1471-2164-9-418. PMC 2562395. PMID 18796152.
- ^ Wheelan SJ, Martínez Murillo F, Boeke JD (July 2008). "The incredible shrinking world of DNA microarrays". Molecular BioSystems. 4 (7): 726–732. doi:10.1039/b706237k. PMC 2535915. PMID 18563246.
- ^ Miyakoshi M, Nishida H, Shintani M, Yamane H, Nojiri H (January 2009). "High-resolution mapping of plasmid transcriptomes in different host bacteria". BMC Genomics. 10: 12. doi:10.1186/1471-2164-10-12. PMC 2642839. PMID 19134166.
- ^ Denoeud F, Aury JM, Da Silva C, Noel B, Rogier O, Delledonne M, et al. (2008). "Annotating genomes with massive-scale RNA sequencing". Genome Biology. 9 (12): R175. doi:10.1186/gb-2008-9-12-r175. PMC 2646279. PMID 19087247.
- ^ Clough E, Barrett T (2016). "The Gene Expression Omnibus Database". Statistical Genomics. Methods in Molecular Biology. Vol. 1418. pp. 93–110. doi:10.1007/978-1-4939-3578-9_5. ISBN 978-1-4939-3576-5. PMC 4944384. PMID 27008011.
- ^ Kiliç S, White ER, Sagitova DM, Cornish JP, Erill I (January 2014). "CollecTF: a database of experimentally validated transcription factor-binding sites in Bacteria". Nucleic Acids Research. 42 (Database issue): D156–D160. doi:10.1093/nar/gkt1123. PMC 3965012. PMID 24234444.
- ^ Moretto M, Sonego P, Dierckxsens N, Brilli M, Bianco L, Ledezma-Tejeida D, et al. (January 2016). "COLOMBOS v3.0: leveraging gene expression compendia for cross-species analyses". Nucleic Acids Research. 44 (D1): D620–D623. doi:10.1093/nar/gkv1251. PMC 4702885. PMID 26586805.
- ^ Faith JJ, Driscoll ME, Fusaro VA, Cosgrove EJ, Hayete B, Juhn FS, et al. (January 2008). "Many Microbe Microarrays Database: uniformly normalized Affymetrix compendia with structured experimental metadata". Nucleic Acids Research. 36 (Database issue): D866–D870. doi:10.1093/nar/gkm815. PMC 2238822. PMID 17932051.
